Earlier, we learned that greenhouse gases are what keep Earth warm. Now let’s look at their natural sources, the ways they are removed (sinks), and how humans are adding extra gases to the mix.
1. Carbon dioxide (CO₂)
Carbon has been cycling between the Earth's surface and the atmosphere for billions of years.
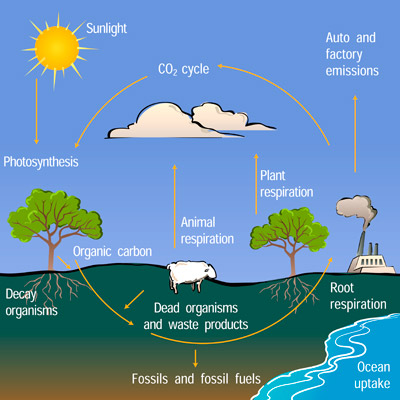
In the atmosphere, carbon exists as carbon dioxide. On Earth, carbon is stored in rocks and sediment, living organisms, and fossil fuels.
Sinks:
- Forests are large carbon sinks. They take out carbon dioxide from the atmosphere through the process of photosynthesis.
- Oceans are also a storehouse of carbon.
Source:
- Carbon dioxide is released into the atmosphere when living organisms die and decompose.
- Oceans release dissolved carbon dioxide directly into the air from the water's surface. It is also released as a result of forest fires and volcanic eruptions.
These natural sources and sinks have been keeping atmospheric carbon dioxide in balance. However, we humans are changing this balance.
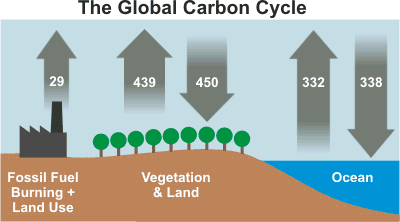
By burning fossil fuels for heat, electricity, and transportation, and activities such as farming and manufacturing, humans release 30-40 gigatons of carbon dioxide each year. Compare this to just 100-300 million tons released by volcanoes each year!
Unfortunately, all this excess carbon dioxide is building up in the atmosphere, where it causes warming.
2. Methane (CH₄)
Methane is naturally released from wetlands, oceans, volcanoes, and termites.
Human activity is responsible for an additional 55-65% (about 500 million tons) of methane emissions each year.
Methane comes from the following sources:
- Extraction, transportation, and burning of fossil fuels
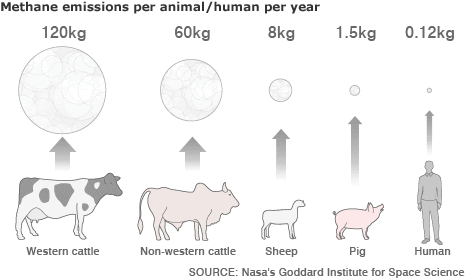
- Livestock farming for meat. Did you know methane is released from the digestive tracts of cattle, pigs, and sheep? (Fig 3.4.3)
- Rice farming, decomposing waste in landfills, and sewage.
3. Nitrous oxide (N₂O )
This gas is released naturally when bacteria in the soil and on plants break down nitrogen in the atmosphere. However, humans use fertilizers and burn fossil fuels. This has increased the amount of N₂O by 20% since 1850.
4. Ozone (O₃)
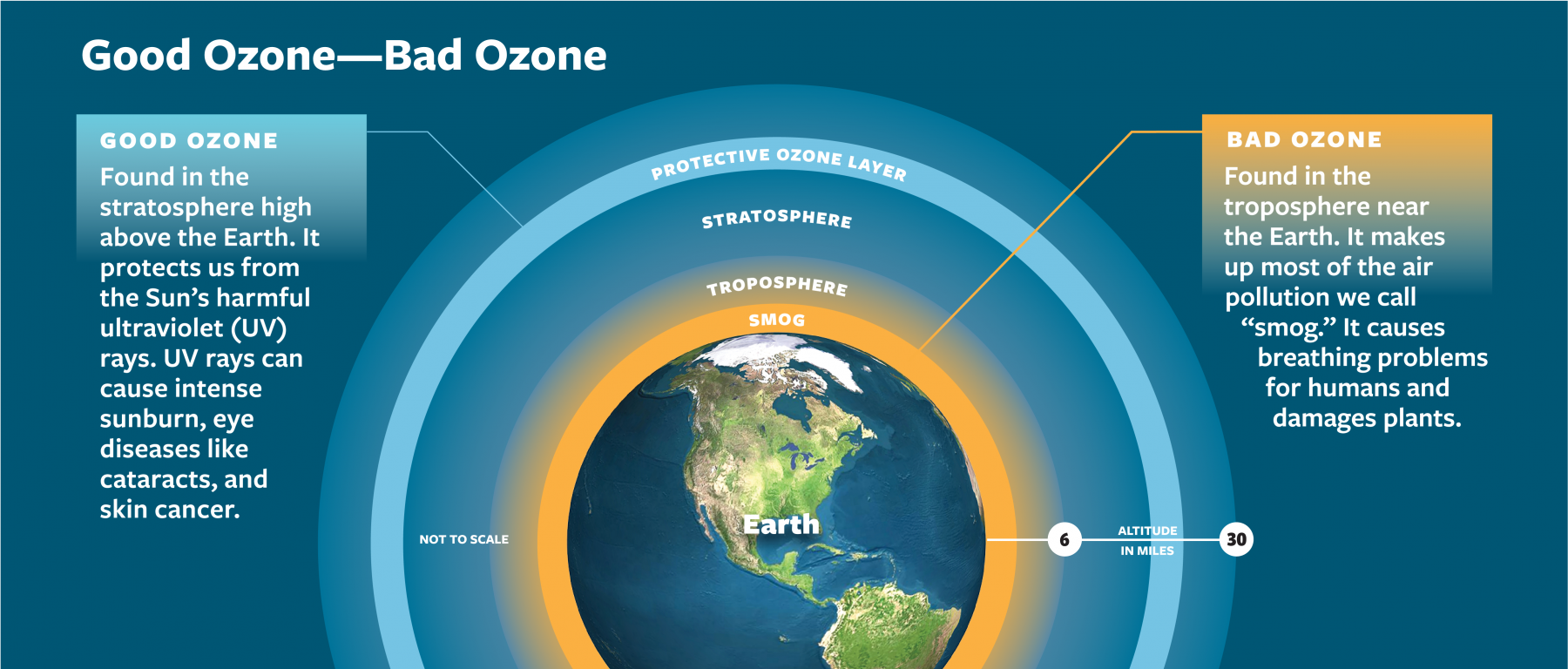
Around 90% of ozone occurs naturally. It lives in the stratosphere, where it traps harmful ultraviolet (UV) rays from the Sun.
However, ground-level ozone has been rising. It is produced when emissions from vehicles and industries release chemicals. These chemicals interact with sunlight to form ground ozone.
Since the late 1800s, ozone levels have gone up by 30% in the lower atmosphere, where it acts as a greenhouse gas.
5. Water Vapor

Water vapor is the most abundant greenhouse gas. It is responsible for 80% of the greenhouse gas effect.
However, unlike other greenhouse gases, the amount of water vapor is related to the warming of the atmosphere. This is because warmer air holds more water vapor that evaporates from Earth’s water sources. Hence, water vapor is not directly linked to human activity.
6. Other Pollutants
Fluorinated gases, dust, and soot are a few other human-made pollutants that trap heat.
Gases that damage ozone have been banned. Unfortunately, some harmful chemicals are still being used in refrigerators, air conditioners, and spray cans.
The burning of fossil fuels also releases aerosols and sulfur gases. These have a slight cooling effect and affect cloud formation.
Let’s learn how long greenhouse gases stay in the atmosphere and how scientists measure them next!
Summary
- Carbon dioxide has naturally cycled through the Earth for billions of years. Humans are upsetting the balance through our use of fossil fuels.
- Methane is mostly generated by the animals that are raised for meat by livestock farmers.
- Water vapor is the largest greenhouse gas but it is not directly linked to human activity.
Earlier, we learned that greenhouse gases are what keep Earth warm. Now let’s look at their natural sources, the ways they are removed (sinks), and how humans are adding extra gases to the mix.
Carbon dioxide (CO₂)

Carbon has been cycling between Earth’s surface and the atmosphere for billions of years.
In the atmosphere, it exists mainly as carbon dioxide, while on Earth it is stored in rocks, sediments, living organisms, and fossil fuels.
- Sinks: Forests are major carbon sinks, absorbing CO₂ through photosynthesis. Oceans are another vast reservoir, drawing in CO₂ from the atmosphere and storing it deep underwater for hundreds of years.
- Sources: When plants and animals die and decompose, CO₂ is released. Oceans release some CO₂ back to the air, and so do wildfires and volcanoes.
For millennia, these sources and sinks kept CO₂ levels in balance. But human activity has upset this equilibrium.
Burning fossil fuels for heat, electricity, and transportation, along with farming and manufacturing, now adds 30–40 gigatons of CO₂ each year. Compare that to only 100–300 million tons from volcanoes! This excess CO₂ accumulates in the atmosphere, trapping heat and warming our planet (Fig. 3.4.2).
Methane (CH₄)

Methane is another powerful greenhouse gas. It is released naturally from wetlands, oceans, volcanoes, and even termites. However, human activities are responsible for an additional 55-65% (approximately 500 million tons) of methane emissions each year.
Major human sources include:
- Fossil Fuels: The extraction, transportation, and burning of coal, oil, and natural gas.
- Livestock farming: Animals raised for meat, such as cattle, sheep, and pigs, release methane through their digestive processes (Fig. 3.4.3)
- Agriculture and Waste: Rice farming, decomposing material in landfills, and untreated sewage also emit methane.
Nitrous oxide (N₂O)
Nitrous oxide occurs naturally when soil and plant bacteria break down nitrogen from the atmosphere.
Human activities, especially the use of chemical fertilizers and the burning of fossil fuels and solid waste, have increased N₂O concentrations by 20% compared to pre-industrial levels.
Ozone (O₃)
Ozone is another heat-trapping gas. About 90% of it is found naturally in the stratosphere, where it forms a protective layer that shields Earth from harmful ultraviolet radiation.
However, ground-level ozone, which is created when vehicle and industrial emissions react with sunlight (Fig. 3.4.4), has risen sharply. Since the late 1800s, ozone levels in the lower atmosphere have increased by 30% where it acts as a greenhouse gas.

Water Vapor

Water vapor is the most abundant greenhouse gas, responsible for 80% of the greenhouse effect. It has been part of Earth’s atmosphere for billions of years.
Unlike other greenhouse gases, water vapor is not directly tied to human activity. But human activities impact water vapor concentrations. This is because water vapor levels depend on temperature, and as air gets warmer, it holds more moisture.
Other Pollutants and Aerosols
Fluorinated gases, dust, and soot are additional human-made pollutants that trap heat. While chemicals that destroy the ozone layer have been banned, harmful substances are still used in refrigerators, air conditioners, and spray cans.
The burning of fossil fuels emits aerosols and sulfur gases, which have a slight cooling effect by reflecting more sunlight into space and influencing the formation of clouds.
Next, we will explore how long these greenhouse gases remain in the atmosphere and the methods scientists use to measure them.
